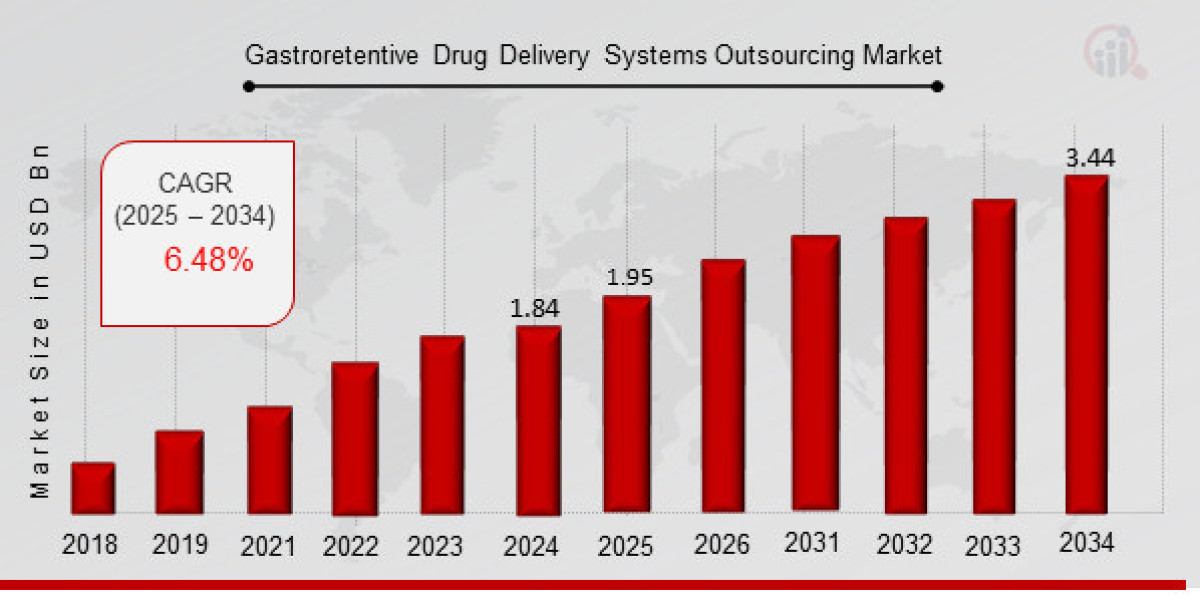
In recent years, pharmaceutical companies have been exploring new and innovative drug delivery systems to address several challenges, including poor bioavailability, irregular drug release, and patient adherence. One of the most exciting and promising areas of advancement is gastroretentive drug delivery systems (GRDDS). These systems are designed to improve the absorption of drugs by prolonging the residence time in the stomach, allowing for a controlled release of the drug. As we step into 2025, the future of GRDDS holds significant promise, particularly in the realm of outsourcing, which is becoming a pivotal force in accelerating innovation, reducing costs, and improving patient outcomes.
To Get More Detailed Analysis of this Market Report Click On Below Link
The Next Revolution in Pharma: What’s Next for Gastroretentive Drug Delivery Systems?
In recent years, pharmaceutical companies have been exploring new and innovative drug delivery systems to address several challenges, including poor bioavailability, irregular drug release, and patient adherence. One of the most exciting and promising areas of advancement is gastroretentive drug delivery systems (GRDDS). These systems are designed to improve the absorption of drugs by prolonging the residence time in the stomach, allowing for a controlled release of the drug. As we step into 2025, the future of GRDDS holds significant promise, particularly in the realm of outsourcing, which is becoming a pivotal force in accelerating innovation, reducing costs, and improving patient outcomes.
What Are Gastroretentive Drug Delivery Systems?
Gastroretentive drug delivery systems are designed to remain in the stomach for extended periods, providing controlled and sustained release of the active pharmaceutical ingredient (API). This system is particularly advantageous for drugs with poor solubility or drugs that need to act in the upper gastrointestinal tract for an extended period. By improving the pharmacokinetics of drugs, GRDDS can enhance therapeutic effectiveness and reduce the frequency of dosing, which in turn improves patient compliance.
Some of the popular technologies for GRDDS include floating systems, mucoadhesive systems, and swellable systems, each designed with different mechanisms to prolong gastric retention. These systems offer various benefits, including enhanced drug absorption, better bioavailability, and a reduction in the dosing frequency.
The Growing Demand for GRDDS in the Pharma Industry
The global demand for gastroretentive drug delivery systems has grown exponentially in recent years, driven by the increasing need for novel drug delivery systems in therapeutic areas like chronic disease management, diabetes, and cardiovascular diseases. As the population ages and the prevalence of lifestyle diseases rises, the demand for medications that offer long-lasting therapeutic effects without the need for frequent dosing is becoming more critical.
Furthermore, GRDDS is proving to be particularly effective for drugs with narrow absorption windows and low solubility. For instance, drugs like metformin, which is used for diabetes management, have a better therapeutic effect when formulated with a gastroretentive system, as it helps maintain the required plasma drug concentrations for longer durations. This is one reason why pharmaceutical companies are investing heavily in the development of GRDDS to expand their therapeutic offerings.
Outsourcing the Development of Gastroretentive Drug Delivery Systems
The development of GRDDS is a complex and resource-intensive process that requires a high level of expertise and access to cutting-edge technologies. While many large pharmaceutical companies may have the infrastructure to handle this in-house, the trend of outsourcing to specialized contract research organizations (CROs) and contract development and manufacturing organizations (CDMOs) has gained significant traction.
Outsourcing GRDDS development offers several benefits, including access to advanced technologies, reduction in production costs, and faster time-to-market. CROs and CDMOs typically have specialized knowledge and state-of-the-art facilities that enable them to quickly scale up the development of GRDDS, ensuring the use of the most efficient and innovative techniques.
For smaller and mid-sized pharmaceutical companies, outsourcing to experienced partners can be a game-changer. It allows these companies to leverage the expertise of specialized firms without having to invest heavily in research and development infrastructure. Moreover, outsourcing enables companies to access global markets and expand their offerings by outsourcing specific tasks such as formulation development, clinical trial management, or manufacturing.
Why Outsourcing is Becoming Essential for GRDDS
Access to Specialized Expertise
Gastroretentive drug delivery systems require a deep understanding of the biological processes involved in drug absorption and release. Outsourcing GRDDS development to specialized CROs and CDMOs ensures that companies can tap into this highly specialized knowledge. These partners have the experience and technological infrastructure to ensure the development of highly effective systems tailored to the specific needs of each drug.
Faster Time-to-Market
The pharmaceutical industry is highly competitive, and bringing a new drug to market requires speed and efficiency. By outsourcing the development of GRDDS, pharmaceutical companies can significantly shorten the development timeline. CROs and CDMOs have the expertise and resources to quickly scale up and manufacture GRDDS formulations, accelerating the timeline from development to market launch.
Cost-Effective Solution
Developing a gastroretentive drug delivery system in-house can be a costly venture, especially for smaller pharmaceutical companies. Outsourcing allows these companies to avoid the significant capital investment in specialized equipment and facilities. By working with experienced outsourcing partners, companies can reduce operational costs while ensuring the highest level of quality control in drug formulation and manufacturing.
Regulatory Compliance and Global Reach
Regulatory requirements for pharmaceutical products can vary significantly from region to region, creating hurdles for companies looking to expand their market reach. Outsourcing to a CRO or CDMO with global experience ensures that GRDDS products are developed in compliance with international regulatory standards. These partners can navigate complex regulatory landscapes, which is crucial for obtaining approval in various regions such as the US, Europe, and Asia.
The Future of Gastroretentive Drug Delivery Systems
Looking ahead, several trends indicate that GRDDS will continue to evolve in exciting ways. The next generation of GRDDS may focus on personalized medicine, with systems that can be tailored to an individual’s unique biology, improving therapeutic outcomes. Additionally, the integration of digital technologies such as smart drug delivery systems, which can monitor and adjust drug release in real time, may revolutionize the market even further.
As the demand for GRDDS grows and the technology continues to evolve, outsourcing will play a critical role in enabling pharmaceutical companies to deliver innovative products to market faster and more cost-effectively. By leveraging external expertise, pharmaceutical companies can continue to make strides in developing safer, more effective, and patient-friendly gastroretentive drug delivery systems.
Conclusion
In conclusion, the future of gastroretentive drug delivery systems is incredibly promising, with exciting technological advancements on the horizon. Outsourcing is becoming an increasingly important strategy for pharmaceutical companies looking to leverage specialized expertise, reduce costs, and accelerate the time-to-market of GRDDS products. As we move into 2025, the pharmaceutical industry is poised for a revolution, with gastroretentive drug delivery systems playing a significant role in shaping the future of patient care and treatment outcomes.
What Are Gastroretentive Drug Delivery Systems?
Gastroretentive drug delivery systems are designed to remain in the stomach for extended periods, providing controlled and sustained release of the active pharmaceutical ingredient (API). This system is particularly advantageous for drugs with poor solubility or drugs that need to act in the upper gastrointestinal tract for an extended period. By improving the pharmacokinetics of drugs, GRDDS can enhance therapeutic effectiveness and reduce the frequency of dosing, which in turn improves patient compliance.
Some of the popular technologies for GRDDS include floating systems, mucoadhesive systems, and swellable systems, each designed with different mechanisms to prolong gastric retention. These systems offer various benefits, including enhanced drug absorption, better bioavailability, and a reduction in the dosing frequency.
The Growing Demand for GRDDS in the Pharma Industry
The global demand for gastroretentive drug delivery systems has grown exponentially in recent years, driven by the increasing need for novel drug delivery systems in therapeutic areas like chronic disease management, diabetes, and cardiovascular diseases. As the population ages and the prevalence of lifestyle diseases rises, the demand for medications that offer long-lasting therapeutic effects without the need for frequent dosing is becoming more critical.
Furthermore, GRDDS is proving to be particularly effective for drugs with narrow absorption windows and low solubility. For instance, drugs like metformin, which is used for diabetes management, have a better therapeutic effect when formulated with a gastroretentive system, as it helps maintain the required plasma drug concentrations for longer durations. This is one reason why pharmaceutical companies are investing heavily in the development of GRDDS to expand their therapeutic offerings.
Outsourcing the Development of Gastroretentive Drug Delivery Systems
The development of GRDDS is a complex and resource-intensive process that requires a high level of expertise and access to cutting-edge technologies. While many large pharmaceutical companies may have the infrastructure to handle this in-house, the trend of outsourcing to specialized contract research organizations (CROs) and contract development and manufacturing organizations (CDMOs) has gained significant traction.
Outsourcing GRDDS development offers several benefits, including access to advanced technologies, reduction in production costs, and faster time-to-market. CROs and CDMOs typically have specialized knowledge and state-of-the-art facilities that enable them to quickly scale up the development of GRDDS, ensuring the use of the most efficient and innovative techniques.
For smaller and mid-sized pharmaceutical companies, outsourcing to experienced partners can be a game-changer. It allows these companies to leverage the expertise of specialized firms without having to invest heavily in research and development infrastructure. Moreover, outsourcing enables companies to access global markets and expand their offerings by outsourcing specific tasks such as formulation development, clinical trial management, or manufacturing.
Why Outsourcing is Becoming Essential for GRDDS
Access to Specialized Expertise
Gastroretentive drug delivery systems require a deep understanding of the biological processes involved in drug absorption and release. Outsourcing GRDDS development to specialized CROs and CDMOs ensures that companies can tap into this highly specialized knowledge. These partners have the experience and technological infrastructure to ensure the development of highly effective systems tailored to the specific needs of each drug.
Faster Time-to-Market
The pharmaceutical industry is highly competitive, and bringing a new drug to market requires speed and efficiency. By outsourcing the development of GRDDS, pharmaceutical companies can significantly shorten the development timeline. CROs and CDMOs have the expertise and resources to quickly scale up and manufacture GRDDS formulations, accelerating the timeline from development to market launch.
Cost-Effective Solution
Developing a gastroretentive drug delivery system in-house can be a costly venture, especially for smaller pharmaceutical companies. Outsourcing allows these companies to avoid the significant capital investment in specialized equipment and facilities. By working with experienced outsourcing partners, companies can reduce operational costs while ensuring the highest level of quality control in drug formulation and manufacturing.
Regulatory Compliance and Global Reach
Regulatory requirements for pharmaceutical products can vary significantly from region to region, creating hurdles for companies looking to expand their market reach. Outsourcing to a CRO or CDMO with global experience ensures that GRDDS products are developed in compliance with international regulatory standards. These partners can navigate complex regulatory landscapes, which is crucial for obtaining approval in various regions such as the US, Europe, and Asia.
The Future of Gastroretentive Drug Delivery Systems
Looking ahead, several trends indicate that GRDDS will continue to evolve in exciting ways. The next generation of GRDDS may focus on personalized medicine, with systems that can be tailored to an individual’s unique biology, improving therapeutic outcomes. Additionally, the integration of digital technologies such as smart drug delivery systems, which can monitor and adjust drug release in real time, may revolutionize the market even further.
As the demand for GRDDS grows and the technology continues to evolve, outsourcing will play a critical role in enabling pharmaceutical companies to deliver innovative products to market faster and more cost-effectively. By leveraging external expertise, pharmaceutical companies can continue to make strides in developing safer, more effective, and patient-friendly gastroretentive drug delivery systems.
Conclusion
In conclusion, the future of gastroretentive drug delivery systems is incredibly promising, with exciting technological advancements on the horizon. Outsourcing is becoming an increasingly important strategy for pharmaceutical companies looking to leverage specialized expertise, reduce costs, and accelerate the time-to-market of GRDDS products. As we move into 2025, the pharmaceutical industry is poised for a revolution, with gastroretentive drug delivery systems playing a significant role in shaping the future of patient care and treatment outcomes.